Updated Covid-19 boosters offer protection, but new studies suggest they don't offer an edge against Omicron | CNN
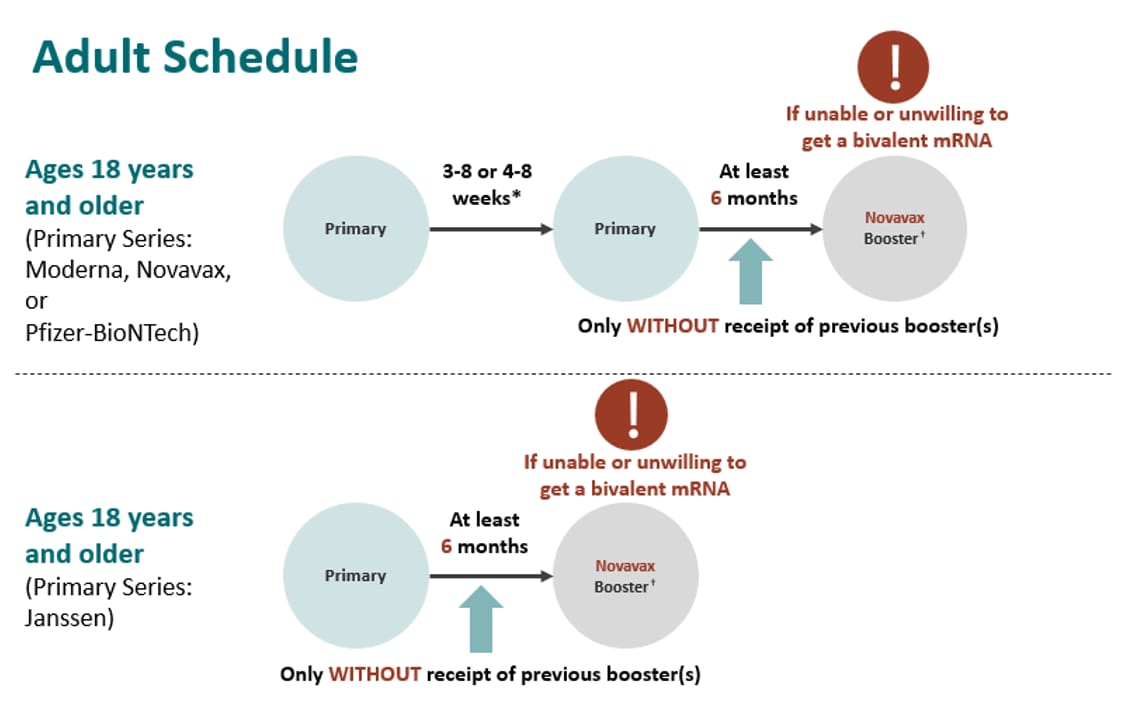
ACIP Evidence to Recommendations (EtR) for Use of Novavax COVID-19 Vaccine Booster Dose for adults ages 18 years and older under an Emergency Use Authorization | CDC