European Medicines Agency Recommends Second COVID Booster For People Over 60 - As WHO Ponders Status Of COVID Emergency - Health Policy Watch

EMA and ECDC: Second booster dose against Covid-19 should be considered for people 60-79 – The Sofia Globe

European Commission - Vaccinate, vaccinate and boost! Evidence gathered by the European Medicines Agency and the European Centre for Disease Prevention and Control (ECDC) suggest that mRNA vaccines used as boosters 3
ECDC-EMA statement on booster vaccination with Omicron adapted bivalent COVID-19 vaccines | CDE Almería - Centro de Documentación Europea - Universidad de Almería

Brianda Echevarria on LinkedIn: EMA evaluating data on booster dose of COVID-19 Vaccine Janssen - European…
Third COVID-19 dose approved by EMA 28 days after second vaccination - Hospital Pharmacy EuropeHospital Pharmacy Europe
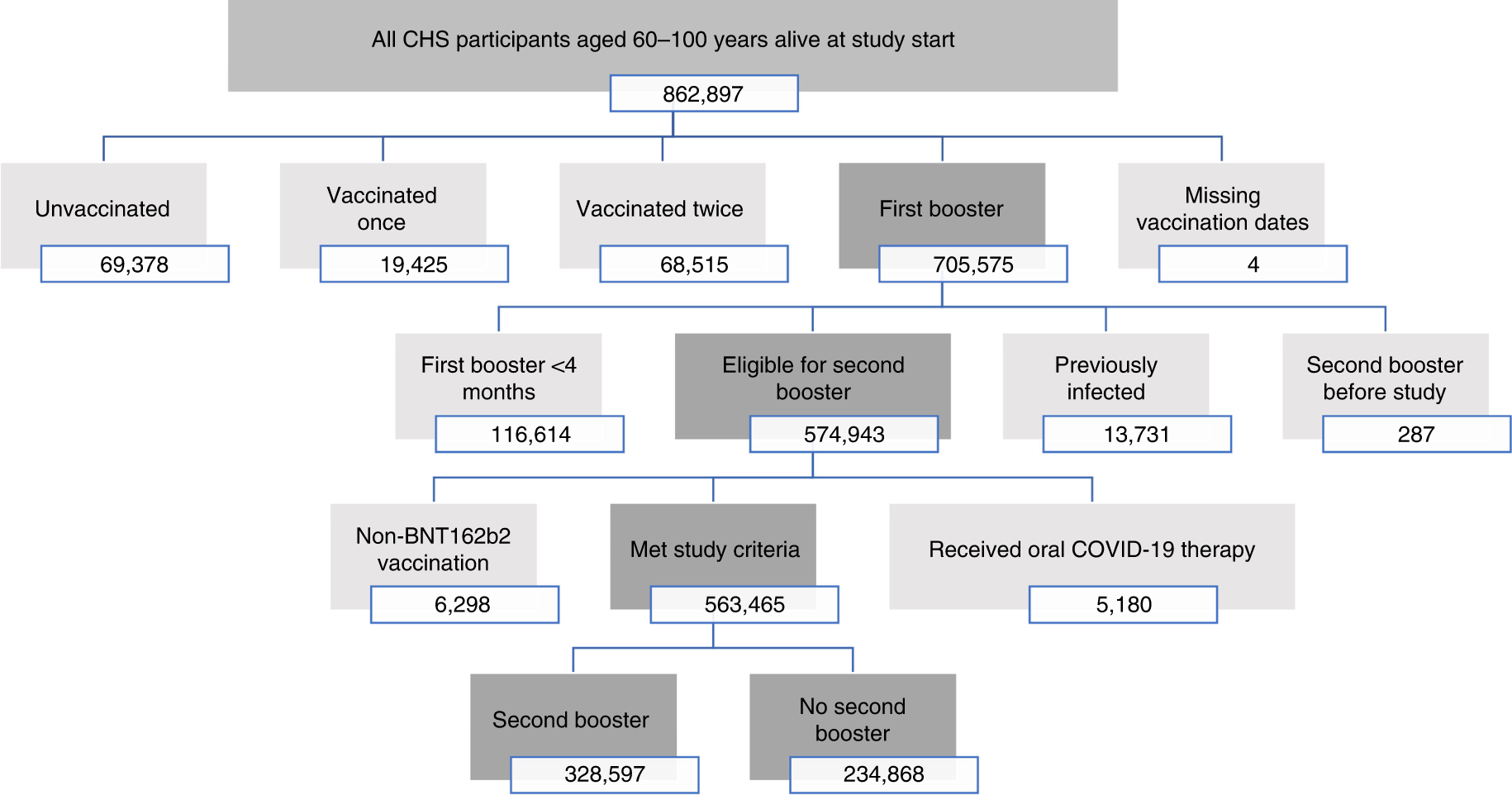
Effectiveness of a second BNT162b2 booster vaccine against hospitalization and death from COVID-19 in adults aged over 60 years | Nature Medicine